Researchers at Texas Biomed and University of Alabama at Birmingham discovered an antibody cocktail against COVID-19 that appears effective against all variants and other coronaviruses. The cocktail has been exclusively licensed to Aridis Pharmaceuticals, which is seeking a manufacturing partner to advance the treatment to human clinical trials.
SAN ANTONIO (March 17, 2022) – As SARS-CoV-2 has continued to evolve and outsmart existing treatments, scientists have not let up looking for ever-more effective tools to keep people safe and successfully recover from COVID-19, and to prepare for future outbreaks.
A long-time partnership between Texas Biomedical Research Institute Professor Luis Martinez-Sobrido, and University of Alabama at Birmingham Associate Professor James Kobie and Professor Mark Walter, has resulted in the discovery of a promising treatment – one that the virus cannot so easily escape.
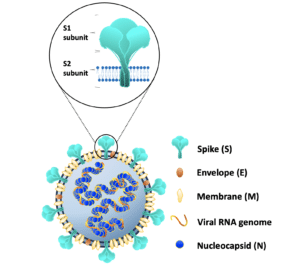
The collaborators found two antibodies that target different parts of the virus’s spike protein and more importantly, block different steps required for viral infection.
“This cocktail has worked against all SARS-CoV-2 variants, including Omicron, as well as SARS-CoV and MERS-CoV – the two coronaviruses that caused past outbreaks,” says Martinez-Sobrido. “Given that, we expect this antibody cocktail to also work against future coronavirus outbreaks as well.”
The researchers recently shared a pre-print of their research paper describing the cocktail. The first antibody targets the top of the spike protein, which helps block the virus from entering a person’s cells. It is similar to other antibody treatments, but continues to work against the latest variants, while others have not. The second antibody targets a lower section of the spike protein, which helps block the virus from fusing with the cell’s membrane and releasing its genetic material into the cell.
This second antibody has the potential to be a game changer because it is targeting a much more stable section of coronaviruses that has been conserved, or remains the same, in SARS-CoV and MERS-CoV through all the variants of SARS-CoV-2, including Omicron. SARS-CoV-2 has continued to evolve throughout the pandemic, as is typical and expected of any virus. Some of those mutations have meant other antibodies can no longer latch on and neutralize the virus. But if SARS-CoV-2 mutates this stable lower section of the spike’s stem, it will not be able to replicate inside its host, and would die out. “It is the Achilles’ heel of the virus,” says Tracey Baas, Innovations Manager at Texas Biomed and manager for the Vaccine Development Center of San Antonio.
The antibodies were found in the serum of recovered COVID-19 patients. The UAB team collected blood samples for research with patients’ informed consent, and screened them for SARS-CoV-2-specific antibodies. The UAB team then replicated the antibodies in large enough quantities so they could be used in multiple studies. Those antibodies were sent to Texas Biomed, where researchers tested them against the virus in biosafety laboratories, first in cells and then in small animal models, which were established or validated at Texas Biomed. The team was able to see the results in cells and animal models in real time using fluorescent viruses they developed.
“We have tested hundreds and hundreds of antibodies since the start of the pandemic,” Martinez-Sobrido says. “But all that effort paid off by identifying these two novel antibodies that target different parts of the spike protein, making for an extremely effective, broad-spectrum treatment.”
UAB and Texas Biomed teamed up with Aridis Pharmaceuticals to develop successful antibody candidates into an inhaled treatment, much like inhalers for asthma. This delivery system promises to be low cost and easy to distribute because it can be self-administered.
“Our researchers used their decades of experience to step back and look for solutions to tough problems,” says Cory Hallam, VP of Business Development at Texas Biomed. “But we are not in the business of manufacturing treatments; we partner with industry to quickly translate discoveries made in our labs into approved products to treat people.”
Texas Biomed is actively working with Aridis to identify potential manufacturing partners that can produce the treatments, so they can move into human clinical trials.
The antibodies have many promising applications, by treating those who fall ill around the world, and by providing antibodies to people who have trouble creating their own even though they have been vaccinated.
“Right now, we don’t have a sterilizing vaccine for COVID-19 like we do for polio, so these antibodies help complete the package of prevention and treatment,” Hallam says.
The Bill and Melinda Gates Foundation recently awarded Aridis, in collaboration with Texas Biomed and UAB, nearly $2 million to develop low cost, inhaled antibody treatments for COVID-19, the flu and other infectious pathogens. Some of that funding will support continued research, development and testing at Texas Biomed, including studies to ensure the cocktail remains effective as new variants emerge.
###
ABOUT TEXAS BIOMED
Texas Biomed is one of the world’s leading independent biomedical research institutions dedicated to eradicating infection and advancing health worldwide through innovative biomedical research. Texas Biomed partners with researchers and institutions around the world to develop vaccines and therapeutics against viral pathogens causing AIDS, hepatitis, hemorrhagic fever, tuberculosis and parasitic diseases responsible for malaria and schistosomiasis disease. The Institute has programs in host-pathogen interactions, disease intervention and prevention, and population health to understand the links between infectious diseases and other diseases such as aging, cardiovascular disease, diabetes and obesity. For more information on Texas Biomed, go to www.TxBiomed.org.